Research Projects
Our challenge is developing new chemical tools to study and manipulate biological systems that have not been tractable.
Project 3. Design and Molecular Evolution of Synthetic Oligomers for Manipulating Biological Functions
Ongoing project 2: Design of peptoid-type synthetic oligomers that are membrane permeable and exhibit high affinity to biopolymers
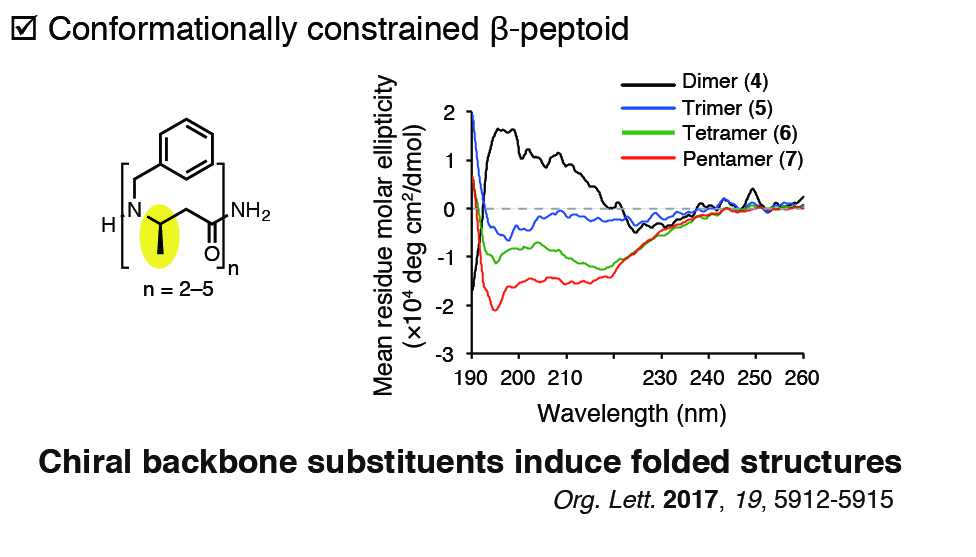
Peptoids (i.e. N-substituted glycine oligomers) are known to be highly membrane permeable peptidomimetic oligomers. However, the conformational flexibility of their backbones prohibits their folding into defined tertiary structures. To overcome the limitation, we have been attempting to produce conformationally-constrained peptoids by introducing chiral backbone substituents to peptoids. We are developing a molecular eveolution technology of such synthetic oligomers to allow facile discovery of membrane-permeable molecular probes for manipulating biological systems.